Summary
Insects use a wide range of tools for orientation, including visual memory, smell, and counting steps. The tricky question is how they combine and compute different kinds of inputs, and whether their methods can help us understand more complex brains or create artificial ones. Michael Gross investigates. Bottom of the article, you will be able to find a simulator link for How ants find their way.
Main Text
Just over 80 years ago, a young biologist in Leiden, in the Netherlands, handed in a PhD thesis that was only 29 pages long and had been written up in a mad hurry, as the author was keen to join a special expedition to Greenland, conducted in celebration of the International Polar Year 1932–33. And yet, this slim thesis, published in a German scientific journal in the same year (Zeitschrift für vergleichen de Physiologie (1932) 16, 305–334) is still being quoted in the scientific literature today.
The student had conducted what one might call mischievous field experiments. He created landmarks around the nest entries of a wasp species known as bee-wolf (Philantus triangulum), such as a ring of pine cones placed on the ground. But once the wasps had grown used to this, he moved the pine cones a few meters away from the nest and observed returning wasps heading straight to the middle of the cone cycle, but frustrated in their expectation of finding their nest entry there.
This mischief-making led to a long and distinguished scientific career, which eventually earned Niko Tinbergen a Nobel Prize (for Physiology and Medicine in 1973), and it highlighted one of the mechanisms by which insects find their way. Even though Tinbergen’s pine cones elegantly and conclusively proved the use of landmarks in insect homing, it turned out to be just one navigation aid of several at their disposal.
Count your steps
For elegant and simple experiments that give clear answers, scientists obviously look for model organisms that orient in simple surroundings. Therefore, orientation researchers have homed in on desert ants, which can achieve surprising feats of orientation in a seemingly featureless environment.
Research over the last two decades using the desert ant Cataglyphis has shown that its most important orientation tool is the so-called path integrator. Essentially, when the ant leaves its nest to go foraging, it counts the steps and keeps a record of how many steps it is away from home at any given time, like a pedometer. In addition, it also records changes in direction.
This method of orientation lends itself to manipulation by mischievous researchers in Tinbergen’s tradition. One just has to lift up the ant and move it a couple of meters, and observe its attempts to find home. Such airlift experiments typically show the ant’s homing attempts shifted by the distance it has been moved, as long as the path integration is the main tool it relies on.
By contrast, attempts to confuse their step counting by introducing corrugated surfaces and even by removing legs have failed, as the group of Harald Wolf at the University of Ulm, Germany, found in field experiments conducted with Cataglyphis fortis in Tunisia (J. Exp. Biol. (2009) 212, 2893–2901). As long as they can walk and stay in touch with the ground, their step-counting appears to be unflappable. In earlier research, however, Wittlinger and colleagues have succeeded in confusing ants by artificially lengthening or shortening their legs (Science (2006) 312, 1965–1967).

Path finders: Humans can use higher cognitive functions to combine information from a range of sources when they’re trying to find their way, but how do insects achieve this? (Photo: Getty Images.)
If animals want to change directions during their foraging, they will also have to record the direction of their steps along with their number, replacing a one-dimensional movement record with a two-dimensional one. For this purpose, they can use the polarization of sunlight as a compass. Even at night, polarized skylight provides navigation help for bull ants in Australia, as Samuel Reid and colleagues at the Australian National University in Canberra have shown (J. Exp. Biol. (2011) 214, 363–370). Similar observations have previously been made with the dung beetle Scarabaeus zambesianus, which can use polarization information at twilight.

Long run: Experimental setup used by Steck and colleagues to show that path integration in desert ants isn’t affected by corrugated surfaces — if the corrugation is removed for the ant’s return, it will still look for its nest in the right place. (Photo: Kathrin Steck.)
Get the vibe
Studying Cataglyphis noda ants in their natural environment at a field site in Turkey, Cornelia Buehlmann, Bill Hansson and Markus Knaden from the Max Planck Institute for Chemical Ecology in Jena, Germany, discovered additional navigational tools in these insects adapted to sparse environments.
Based on the observation that leaf cutter ants can use vibration signals to call for help when they are trapped, and on recent evidence that suggests the presence of magnetosensation in ants, PhD student Buehlmann tested whether desert ants can use either vibrational landmarks or magnetic ones to find their nest. To the researchers’ surprise, Cataglyphis noda can use either of these easily (PLoS ONE (2012) 7, e33117).
They could train ants to identify their nest by a vibrating mechanism buried near it, and created conflicts between this landmark memory and the path integrator to prove that the ants do trust the vibration more than their step count. To rule out any electromagnetic effects from the mechanism, the researchers also performed a control experiment where the same instrument was as close to the nest but not in contact with the ground.
Similarly, in the magnetism experiments, they could train Cataglyphis noda to recognise their nests by the nearby presence of a strong magnet, which was 500 times stronger than the ambient Earth magnetism. “This doesn’t mean that ants have a kind of sensory organ for the detection of magnetic fields,” Knaden cautions. “Their behaviour could also be caused by abnormal neural electrical signals due to the strong magnetic field which were memorized by the ants.”
In a separate project conducted with the related species Cataglyphis fortis in Tunisia, the Max Planck researchers identified a third novel navigation tool, namely the recognition of the carbon dioxide plume emanating from an active ants nest. However, as this would be the case for any ants nest, and as it is important for the ants to find their own (lest they get killed by the resident colony), this navigation help is only useful in combination with other tools, such as the path integrator (Curr. Biol. (2012) 22, 645–649). The researchers found that the carbon dioxide plume only attracts ants to their nest if the path integration suggests they are in the right area. The research suggests that ants can use any of a wide range of sensory signatures associated with a place of interest to complement their path integration information.
Feel the force
If ant researchers want to find out just how the insects sense magnetic fields, they may look at the precedent of avian magnetosensation, which has proven a big challenge for the last four decades. Ever since Wolfgang Wiltschko discovered the role of magnetic fields in avian orientation in the 1960s, researchers have tried to track down the ‘compass’, but initially with limited success.
One of the two most promising hypotheses is that the bird compass is a chemical reaction using radicals. In 2008, Oxford chemists Christine Timmel and Peter Hore, with their colleagues, reported the first chemical system based on radicals that is sufficiently sensitive to the very weak magnetic field of our planet to serve as a compass. The leading candidate for the as yet elusive real-life version of this chemical compass is the family of cryptochromes, proteins of otherwise unknown function located in the birds’ eyes.
Monarch butterflies possess two different cryptochromes, one resembling that in Drosophila, and another that is more like those of vertebrates. Expressing each of these genes in a cryptochrome-deficient strain of Drosophila, the group of Steven Reppert at the Massachusetts Medical School in Worcester found that each can restore the light-dependent magnetosensitive behaviour of the fly (Nature (2010) 463, 804–807). Intriguingly, the effect was resistant to mutation of a tryptophan residue that other groups expected to be involved in the radical pair mechanism, so the precise molecular mechanism responsible for cryptochrome-dependent magnetosensing remains to be determined.
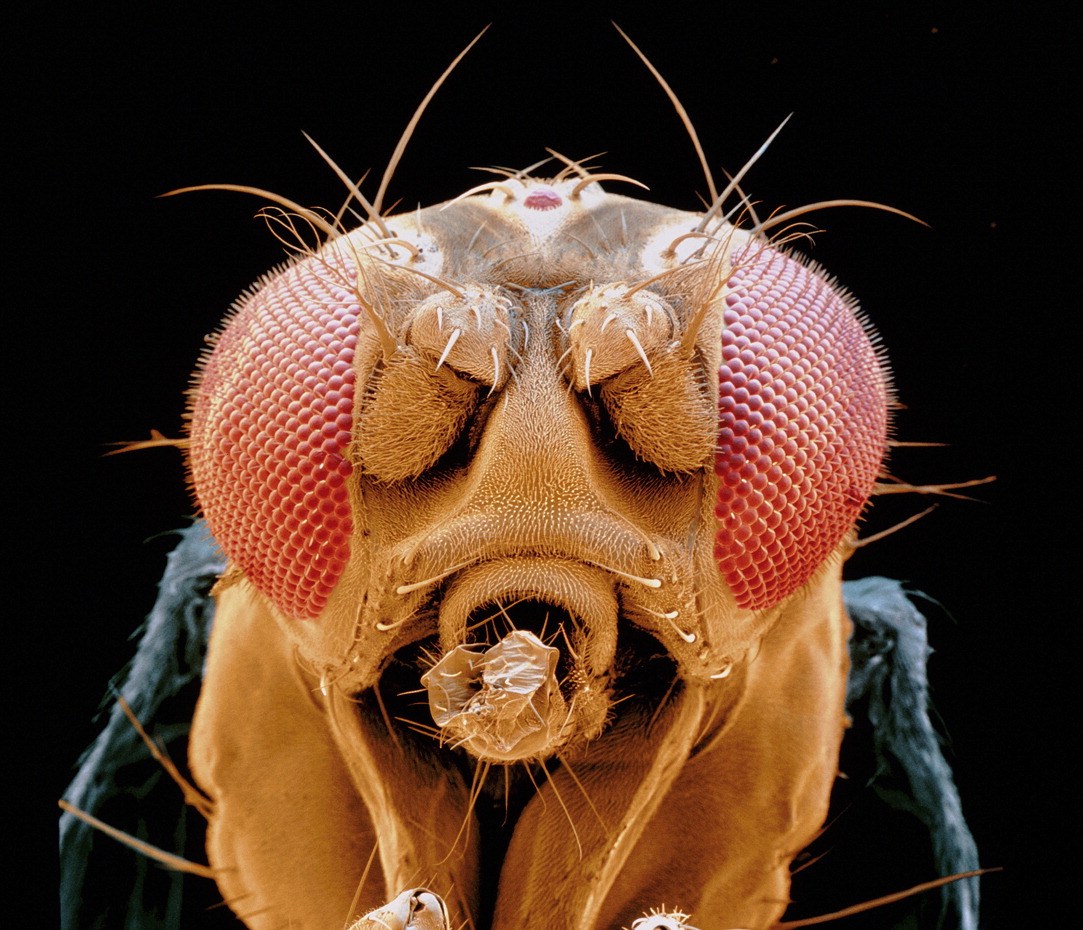
Vision thing: Perception of light plays several roles in insect navigation, including the input of compass information necessary for path integration, and the recognition of visual landmarks. (Photo: Science Photo Library.)
In March 2012, Timmel, Hore and colleagues could report magnetically sensitive photochemical reactions in a plant analogue of the bird cryptochromes that are suggested to lie at the heart of avian navigation (Proc. Natl. Acad. Sci. USA (2012) 109, 4774–4779). Specifically, the authors show that the kinetics and quantum yield of the formation of a flavin–tryptophan radical pair in cryptochromes respond sensitively to magnetic fields of the strength of the Earth’s field, adding further support to the hypothesis that cryptochromes are indeed the molecular compass of migratory birds.
“We are very excited about this result, which demonstrates that cryptochromes are fit for purpose as chemical magnetoreceptors,” says Timmel. “It is now crucial to investigate if such magnetosensitivity can also be observed in cryptochromes from animals known to use the Earth’s magnetic field for navigation.”
Follow your nose
Odours are another very important means of orientation for animals including insects. In their field experiments described above, for instance, Buehlmann and colleagues included controls using olfactory or visual landmarks instead of the magnetic and vibrational landmarks they introduced as novel features. Of all the methods used, the olfactory marking (methyl salicylate in hexane) narrowed the nest search of the ants most efficiently.
However, as Mark Willis from Case Western Reserve University in Cleveland, Ohio, has explained in a recent review (Navigation (2008) 55, 127–135), tracking an odour plume is a complex computational task, as the distribution of the relevant substance in the fluid may be irregular and will depend on flow dynamics. Insects are good models to peel apart these complexities, as many will both fly and walk, i.e. move with the medium carrying the odorant or independently of it. Willis says: “Because the insects we study have complex brains that we are only beginning to understand, it is very important for us to know sensory inputs (i.e., odour plume structure) they experience while tracking odours. By carefully associating the olfactory behaviors we observe with known odour inputs, we can begin to understand how the bug’s brain uses the odour information to find the source emitting the odour. “
In a study using Drosophila in a wind channel, the group of Mark Frye at the University of California at Los Angeles could demonstrate that the flies can perceive substance gradients in an odour plume ‘in stereo’, enabling them to navigate towards a source of food odours, for example (Curr. Biol. (2009) 19, 1301–1307.
Look around you
Unlike the desert ants, most land animals live in environments that will enable them to use visual landmarks for orientation, beyond the use of polarisation or panorama that is necessary to provide compass information in path integration. Ants living in forests, for instance, use path integration only to return to the wider area of their nest and will then use landmarks for the final stretch.
Research into how animals perform navigation by sight has a long history (including, again, Tinbergen’s pine cones), but researchers still have crucial questions to address right now, especially if they want to understand the neuronal basis of the observed behaviour.
“Quite generally, we are just starting to acquire, develop and apply tools that allow us to reconstruct the navigational information available to animals under natural conditions,” says Jochen Zeil from the Australian National University at Canberra. “It is an exciting time and in the next few years, a combination of these reconstruction methods with clever experiments and neurally realistic modelling will make huge advances in our understanding of navigational mechanisms and neuro-computational implementations.” The advantage of insects, as Zeil points out, is that one can manipulate their natural behaviour within their natural environment in ways that wouldn’t be possible with mammals or birds.

This way: Migratory birds use the Earth’s magnetic field for navigation, but the precise nature of their ‘compass’ has remained elusive. New results add weight to the hypothesis that radical reactions are at the heart of it. (Photo: Getty Images.)
In a recent review of visual homing in insects (Curr. Opin. Neurobiol. (2012) 22, 285–293), Zeil also mentions a few of the open questions in the field, including whether or not visual orientation requires the recognition of discrete objects, as opposed to more general panoramic impressions.
“In many experiments, indoors and out, insects clearly respond to individual objects. However, it is not clear what constitutes salient objects under complex natural conditions, because this will depend on both their salience in terms of the signals they provide against a noisy background and on the way information is extracted by the visual system of animals,” Zeil explains. “We know that both localization of goals and direction of heading along routes can be achieved without segmentation of the visual scene into discrete objects, which is considered to be computationally demanding — but who knows?”
The biggest challenge is to find out how the insects store complex geographical information in their very small brains. “There are wonderful anatomical studies showing that certain parts of the brain of insects reconfigure themselves when exposed to information related to navigation,” says Zeil. For instance, recent research demonstrates that the neuroanatomy of ants changes when they are exposed to light for the first time in their life (Dev. Neurobiol. (2010) 70, 408–423). “So the question is,” concludes Zeil, “will we be able to exactly map the available visual and motor-state information to the topography of these changes in the brain?”
Where do we go now?
Another important question is: what are you going to do if you’re driving and the SatNav says one thing, your memory of the last trip says something else, and the person in the passenger seat has a different opinion altogether? We may have more or less rational ways of evaluating and combining information from different sources, but how do ants manage that?
This question, again, calls for mischievous manipulations, by which researchers can create conflicts between the separate navigation systems and can then observe how the problems are resolved, as Matthew Collett from the University of Exeter has recently reported in this journal (Curr. Biol. (2012) 22, 927–932). His observations show that ants can use visual memory and path integration at the same time and may steer in a compromise direction if the two systems give conflicting results. Modelling shows that the observed behaviours can be explained by simple superposition of the outputs of the separate systems. “Insects show how much can be done with a relatively small brain and, through their limitations, suggest which processes really do require larger brains,” Collett concludes.
Insect navigation is important for a whole range of reasons. For neuroscience, it offers the chance to observe information processing in manageable systems under conditions that are close to nature and thus relevant to evolutionary considerations. The crucial importance of navigation and homing behaviours is evident from the observation that most animals need these abilities to some extent — even sessile animals often have navigating larval stages.
Therefore it is no big surprise that evolution has driven natural navigation systems to a degree of perfection that human engineers can only dream of. “I think that every animal we look at is a more competent, more robust, more flexible, more miniaturized and a more energy-, material-, sensor- and computation-efficient agent than anything we have ever built,” concludes Zeil. “So would anyone need more justification for how fundamentally important, intellectually challenging and promising it is to conduct research into the navigational abilities of insects?”
Simulator
To get more details picture about this topic, here we got a nice simulator from Github Pages. Credits goes to the Developer.
GitHub Repo: https://github.com/sveinnthorarins/ant-simulation
Live link: https://sveinnthorarins.github.io/ant-simulation
Credits: sciencedirect.com






